this feels like a potentially sincere attempt to recruit people into an anti-science conspiracy movement - this doesn't really feel different than the kind of reasoning you see with moon landing denialists or flat earthers.
Eh I wouldn't take it too seriously, I'm pretty sure it's a play on the whole running joke of "saying something ridiculous, then end it with 'You guys don't seriously believe this right?!?'" type of thing. I've seen many of these greentexts that used that format recently.
It's kinda funny to me because it loosely reminds me of same logic as those old rage comic "troll physics" memes like these:

Poe's Law
... Old?
Rude.
I just realized I called myself old too :(
2012 was only 7 years ago, right?

2012 is at least 11 years in the future, I'm pretty sure.
It was seven years ago, seven years ago.
And /r/the_donald was just a joke
It's actually not a bad question, just one people don't really think about. Why does room temperature water ~~sublimate~~ evaporate?
It's because the temperature is an average, and some molecules at the surface have enough energy to break their polar bonds.
Water doesn't sublimate. Sublimation is solid to gaseous phase change.
Fixed
sublimation is poorly defined in our context.
Technically, water does sublimate, just not at normal earth pressures. Below 0.6 kPa it transitions straight from solid to gas.
Pretty sure Bill Nye taught me this. Substitute teachers aren't playing the good stuff anymore
I wanna say Bill Nye had a little contraption that explained this phenomenon. A cup with a piston on one end that vibrated. The top part of the cup had a ring in the center where little balls in the cup could fit. The piston represented the temperature (energy). Even at a lower temperature, some balls could randomly fly into the little hole and into the other partition. Turning the temperature up (increasing the speed and power of the piston) made more balls more frequently "evaporate." I wish I could find that demonstration again.
Maybe you just gotta piston pound your balls for yourself, comrade.
Im a lifelong flat earth denier
The oceans aren't carbonated therefore flat earth
Not carbonated enough yet
flat earth is pushed by the global elite pedophiles, after all - it's what they want us to believe
Enter vapor pressure:

Basically water always evaporates if the air is completely dry, until the air contains a certain amount of water (measured in partial pressure, which is the part of the air pressure that is caused by water vapor). This partial pressure is temperature-dependent, so if you have 20°C (normal room temperature) you're gonna have 23 mbar of water vapor partial pressure in the air. Source
So water still evaporates at lower temperatures when the air is dry enough. It's just that at 100°C ("boiling point of water"), that partial pressure of water vapor in the air increases to 1013 mbar which is equal to the total pressure of the air; In other words, at that temperature in equilibrium, the air is totally made up of water vapor and nothing else. If you increase the temperature above that, the water vapor partial pressure tries to still increase, which makes the total pressure go above normal air pressure, which causes a pressure gradient and causes the air to move with mechanical force, which you can use to make turbines spin.
A more microoscopic explanation is due to Maxwell-Boltzmann distribution.
First, you need to underestand temperature. The difference between cold and hot water is the average speed at which particles move, with hotter water's particles moving faster.
But this is just the average speed, it turns out that particle's speed can be se en as a random variable, and they follow Maxwell-Boltzmann distribution:
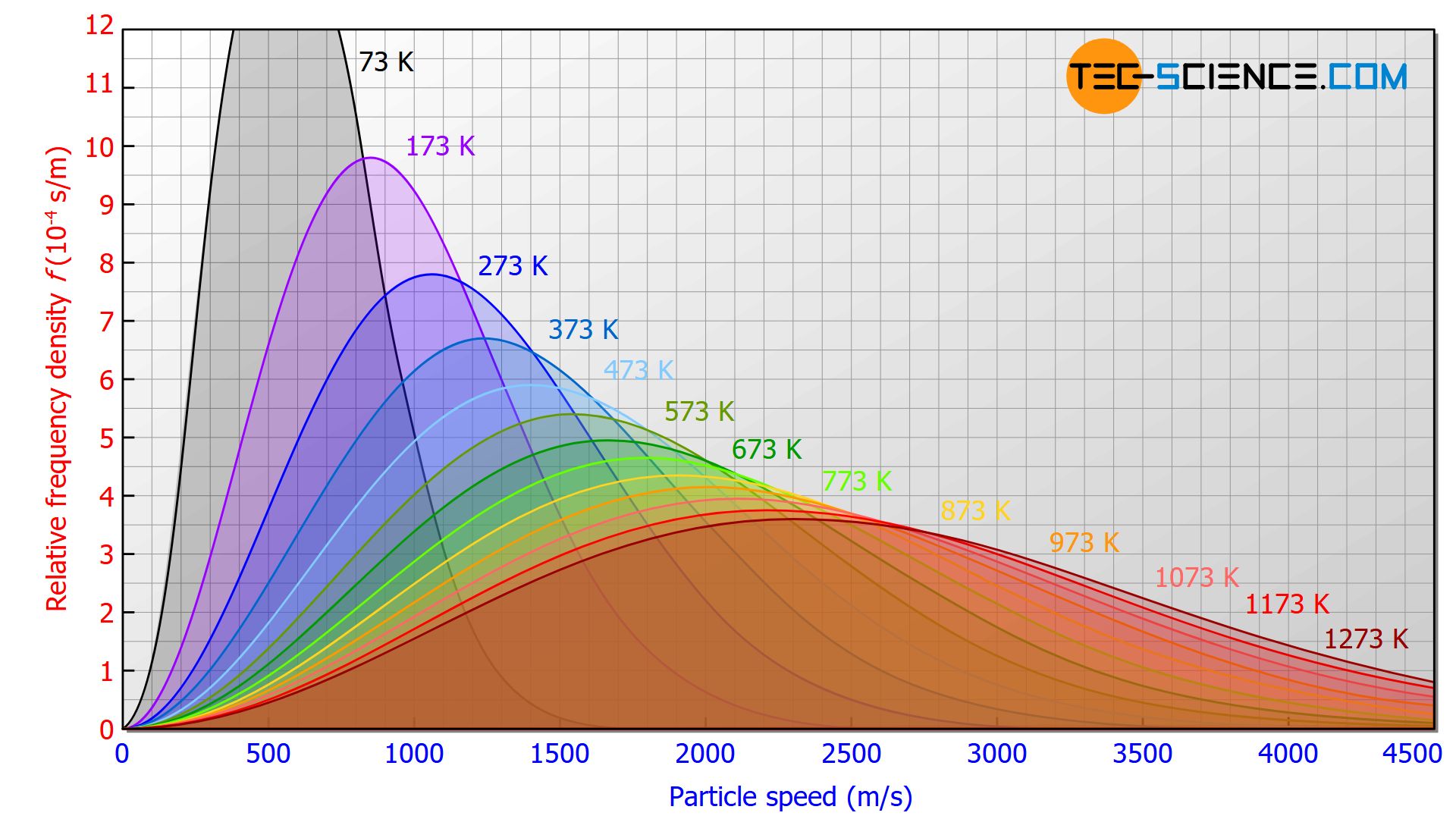
So you have a small proportion of particles that move very fast, even in cold water. If some of those particles get (or collide with other particles near) to the "layer" of water that is on contact with the air, they will have enough energy to escape water's superficial tension, thus going into the air and out of the water body. The higher the average speed of the particles, the faster this process will go. Finally, the rate at which this process happens also depends on the energy required to be able to leave the water body, which depends on factors like air pressure.
yeah i've of course heard about it and i'm studying physics myself rn so i'll get to it.
I simply haven't taken the course on quantum physics yet so i don't want to make bold claims here. I have yet to derive the classical phenomena from quantum physics myself.
You won't see this on a quantum mechanics class, but on my favorite one, statistichal mechanics.
Thank you ive always wondered about this but never wondered about it when I had the chance to look it up and now I know :)
You're welcome :)
You're really good at explaining stuff.
thanks :D
Yeah sure, whatever, nerd.
matter changes state based on temperature
This is a gross oversimplification and your experiment proves that :)
The teacher was explaining about conducting and not conducting, and we had a battery with lamp thing to test on various objects. I of course had to test this on a pencil and discovered semi-conducting. That was a serious "not today" sigh from the teacher.
Everything is a conductor if your generator has a high enough frequency.
Anon still lives with his mom and she mopped up the spill.
It's interesting because very pure water without asperites can be heated above 100c at standard pressure at sea level without boiling. But once impurities are added to it it starts boiling vigorously!
Sounds like the same thing that happens at 0°C with very pure water that stays liquid, but shake it and bam! It insta-freezes.
Sometime water bottles display this trait in temps just below freezing. Liquid water in the bottle but break the seal and pop! Frozen
Doesn't have to be pure for that! I've seen it happen with those cheap plastic sleeve popsicles. It's kind of fun flicking them and watching the ice radiate through.
What are asperites?
Imperfections in the surface of the container the water is in. Sorry, I intended to write 'water in a container without asperites'!
Temperature is based on "average" kinetic energy so technically there are molecules with higher energy's that are higher than the transition state and that's why
I always panic a little, envisioning the vast game of pinball we’re constantly wading through, and also not because the sphere atomic model is just shorthand
For fun I'm going to explain why. So air can hold some amount of water in it at any temperature. Water energy in form of heat is not evenly distributed so some molecules move faster than others, at the surface some move fast enough to escape the water and into the air. That's called evaporation, boiling does however require 100°C.
This also explains why humid weather affects evaporation (lower capacity to hold water in air) and at high humidity there's an a similar chance of water being deposited to the body of water as water escaping which affects evaporation speed a lot.
Honestly, I really like this quality of water, it would be super annoying to deal with otherwise.
Anon didn't pay attention in physics class.
You claim water boils at 212⁰
Yet this sponge soaks it right to at room temperature.
Curious.
"Water can only fly when its a gas!!!" Words from losers that have never looked at the sky.
If water turns into gas at 100 degrees, how come I can pour it into a mug?
Usually, when pouring, it doesn't exceed 90 degrees.
I had this question in a mid-term in physical chemistry 20 years ago. I can't remember the details but it is driven by the entropy in the system. Along with the things other have said about changes right above the surface of the puffle.
Anon forgot that temperature in a substance is not uniform. This normally doesn't matter, but if a part becomes hotter than the boiling point it will leave before it has a chance to go back to average temperature. So yes, the water "went to 212" before evaporating.
Those are the little water goblins stealing your moisture while nobody's looking.
Greentext
This is a place to share greentexts and witness the confounding life of Anon. If you're new to the Greentext community, think of it as a sort of zoo with Anon as the main attraction.
Be warned:
- Anon is often crazy.
- Anon is often depressed.
- Anon frequently shares thoughts that are immature, offensive, or incomprehensible.
If you find yourself getting angry (or god forbid, agreeing) with something Anon has said, you might be doing it wrong.
